 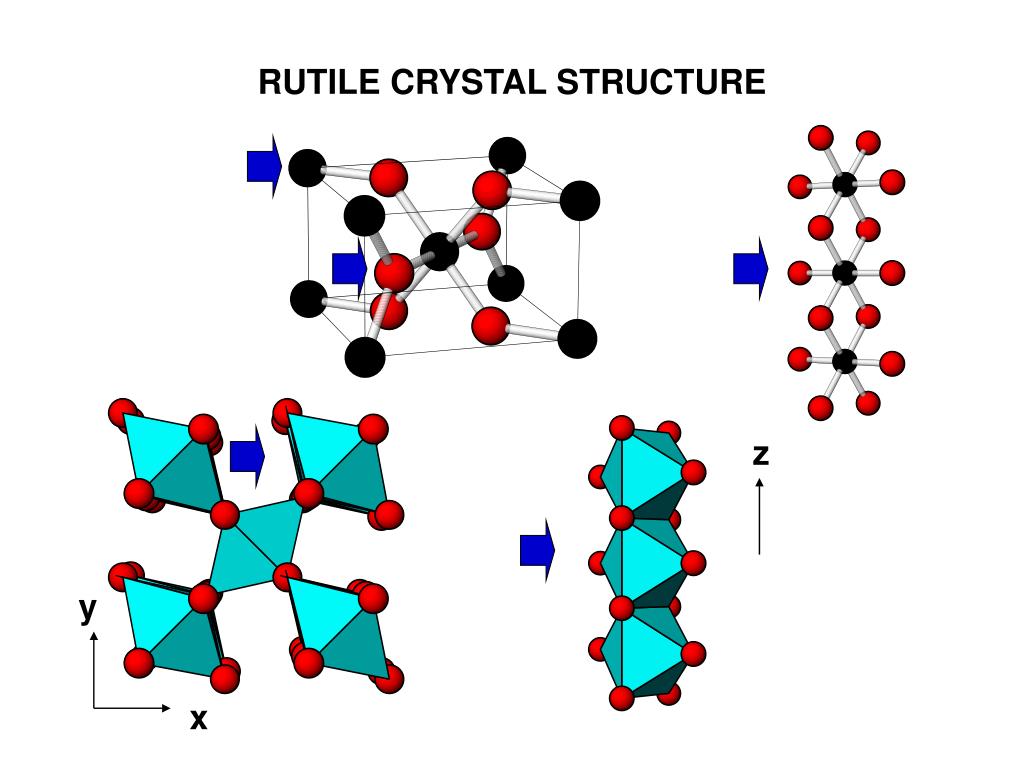
GeO 2 is dimorphous at ambient atmospheric conditions, represented by both rutile-type and α-quartz-structured phases depending on the temperature, but with increasing pressure the GeO 2 rutile becomes more stable, and is the primary phase above two GPa (Micoulaut et al., 2006 ). The GeO 2–TiO 2 phase diagram at elevated pressures and temperatures has not been studied in great detail and the mutual solubility of Ge and Ti in the phases stable at these conditions is still largely unknown. Additionally, at ambient pressure GeO 2 and TiO 2 exhibit only limited mutual solubility. A metastable α-quartz-type structured GeO 2 has also been reported as the result of the cooling of the β-quartz-type structure (Sarver, 1961 ). 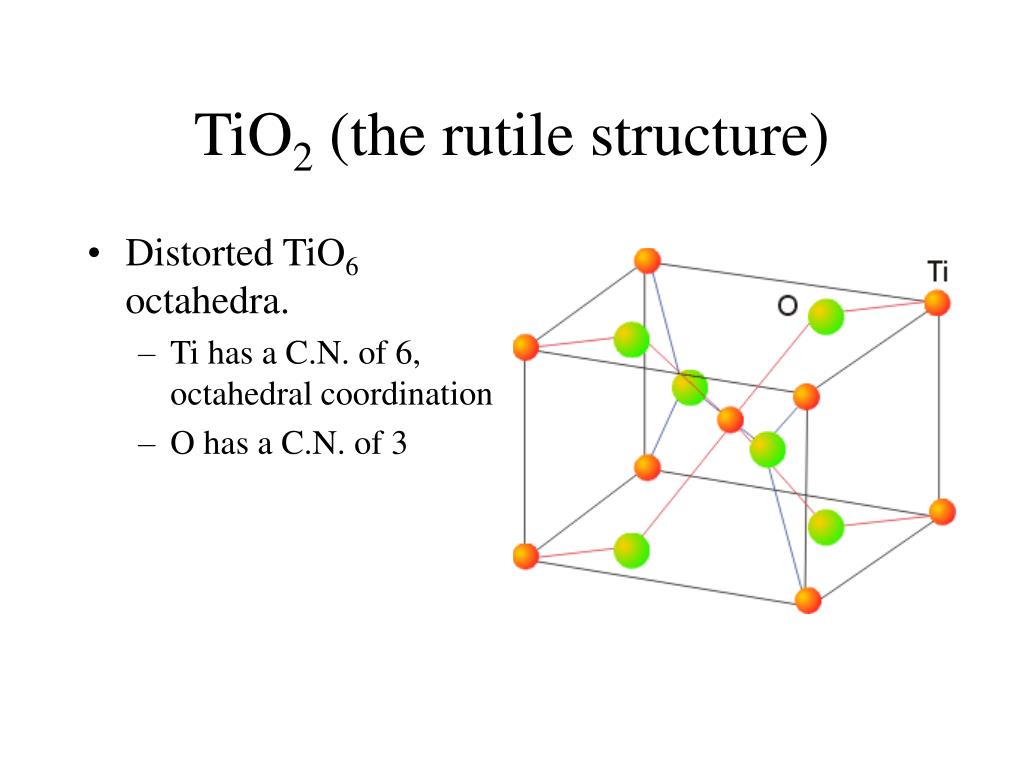
These attractive catalytic properties of Ru xCo 1− xO y can be attributed to the relatively large surface area of the tubular morphology and the substituted structures, presenting feasibility as a practical and economical OER catalyst.At ambient pressure, the GeO 2–TiO 2 phase diagram shows the formation of three phases: rutile-type GeO 2, stable up to 1323 K, β-quartz-type GeO 2, stable above 1323 K and TiO 2 in the form of rutile. Even after more than half of the noble/active Ru content was replaced with cheap/less-active Co, Ru 0.47Co 0.53O y showed a good OER activity and a greatly improved stability compared to RuO 2 under the continuous OER. Ru xCo 1− xO y nanotubes with x ≥ 0.47 presented an excellent OER activity comparable to pure RuO 2, known to be the best OER catalyst. The as-prepared Ru xCo 1− xO y nanotubes were investigated for oxygen evolution reaction (OER) electrocatalytic activity in 1.0 M HClO 4 aqueous solution. This elemental substitution created oxygen vacancies in the rutile structure and also resulted in the incorporation of Ru 3+ in the octahedral sites of the spinel structure. Ru xCo 1− xO y nanotubes (0 < x < 0.77) were composed of both rutile (Ru in RuO 2 is replaced with Co) and spinel (Co in Co 3O 4 is replaced with Ru) structures. A variety of binary Ru–Co mixed oxide nanotubes (Ru xCo 1− xO y with x = 0.19, 0.33, 0.47, 0.64 and 0.77) were readily synthesized via electrospinning and subsequent calcination. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
